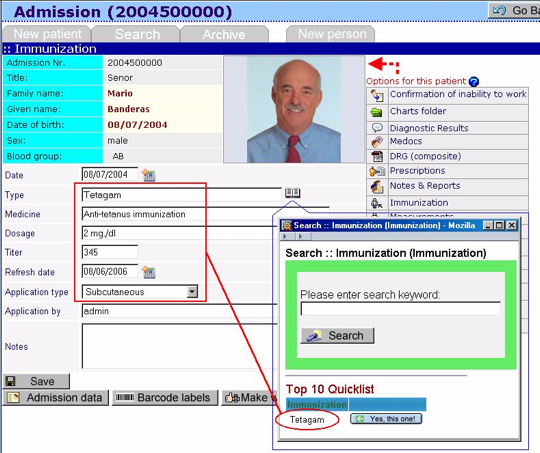
Global regulatory requirements for medical devices. For medical devices. Medical devices can in turn be regulated as one group or regulated separately, usually as one of the subgroups. In europe general medical devices are divided into non invasive devices, invasive devices and active devices. An active medical device is a device that requires a source of energy to function. Dermatology electronic records find top results. Directhit has been visited by 1m+ users in the past month. Technical file vs. 510(k) vs. Design history file what. We’d like to address three documents that cause a lot of confusion and tearing out of hair; the technical file, 510(k) submission and the design history file. As with any kind of files in medical device development, these require a lot of effort, however, if you’ve done the background work, you will find that the information required inter. Design history file (dhf) vs. Device master record (dmr) vs. The device history record is literally the history of the device. Everything that you complied in the dmr was used to make the device. The history and information on how you made the device in accordance with the dmr is stored in the dhr. Much like the dhf is the history of the design, the dhr is the history of the device. Dhf vs. Dmr vs. Dhr. Product documentation (technical file) example medcert. Class i medical devices (conformity assessment) class i (steril or with measuring function) medical devices; class iia medical devices (conformity assessment) class iib medical devices (conformity assessment) class iii medical devices; medical device directive 93/42/eec; iso 13485. What is iso 13485? Risk management. Health record video results. Find health record if you are looking now. Health record welcome to internetcorkboard. Looking for dermatology electronic records? Search now on msn. U.S. Fda medical device master files registrar corp. A medical device master file is a submission of information to the u.S. Food and drug administration by the medical device master file holder. A medical device master file is a submission to u.S. Fda that may be used in support of premarket submissions to provide confidential detailed information about establishments, processes, or articles.
The tool to manage your medical devices dhf aligned.Ch. Reduce the time spent on medical device documentation by 61%. Learn how! Health record definition of health record by medical dictionary. Everymanbusiness has been visited by 100k+ users in the past month. Health record selected results find health record. Healthwebsearch.Msn has been visited by 1m+ users in the past month. Medical device quality systems manual a small entity. Producing a medical device. Manufacturers of medical devices commonly find that their quality needs are broader than these basic elements because of the additional need to meet company quality claims as required by paragraph 501(c) of the federal food, drug, and cosmetic (fd&c) act and to meet customer needs and requirements. Regulation (eu) 2017/ 745 of the european parliament and of. Definition of a medical device or are covered by this regulation. (12) cer tain groups of products for which a manufacturer claims only an aesthetic or another nonmedical pur pose but which are similar to medical devices in terms of functioning and r isks profile should be covered by this regulation. Montgomery county health department. Get more related info visit us now discover more results.

Ehr Patient Dashboard
Directhit has been visited by 1m+ users in the past month. Montgomery county health department our mission to promote, protect and improve the health and prosperity of people in tennessee naloxone training, certification, and free kit available every 3rd wednesday of each month, from 530p.M. 600p.M. At civic hall in the veteran's plaza. The terms medical record, health record, and medical chart are used somewhat interchangeably to describe the systematic documentation of a single patient's medical history and care across time within one particular health care provider's jurisdiction. Master files fda. Master files help preserve the trade secrets of the ancillary medical device industry and facilitate the sound scientific evaluation of medical devices. July 2016 iso 134852016 frequently asked questions. Clause 4.2.3 medical device file what type of file does “medical device file” refer to? “Medical device file” refers to both the device master record, and the technical documentation (technical file or design dossier). The requirements of this clause were previously documented in clause 4.2.1 in iso 134852003 and en iso 134852012. Dermatology electronic records find top results. Only you or your personal representative has the right to access your records. A health care provider or health plan may send copies of your records to another provider or health plan only as needed for treatment or payment or with your permission.
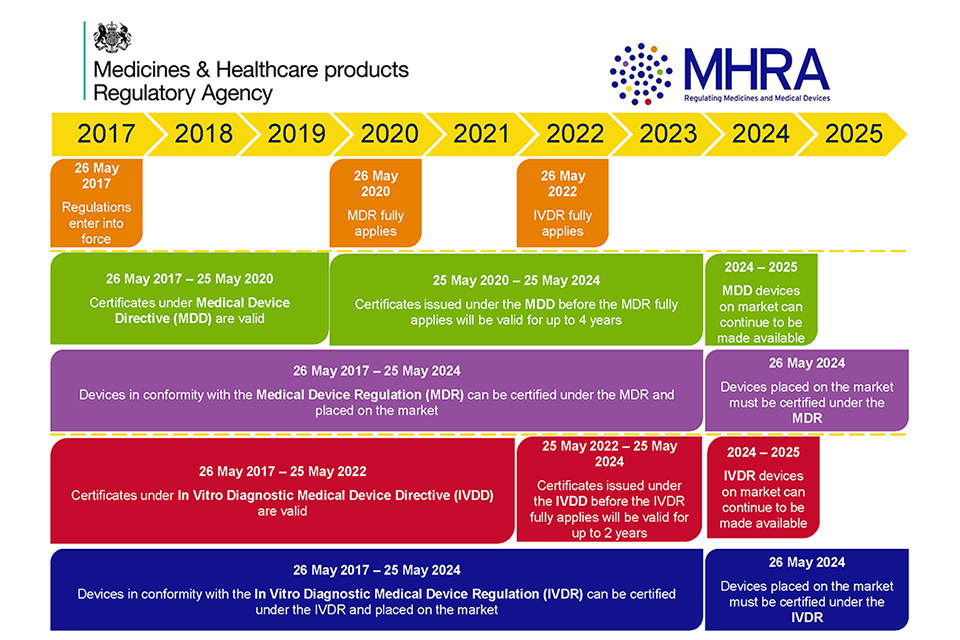
An introductory guide to the medical device regulation (mdr. The european databank on medical devices (eudamed), is a database that those who manufacture and supply medical devices, as well as notified bodies, health institutions and competent authorities, will have access to. Using this system, those involved in manufacturing and supplying medical devices and ivds will. Healthcare records. Healthcare records govtsearches. Search for health records online at directhit. Ce technical files for medical devices emergobyul. Want to sell your medical device in europe? If the answer is yes, you probably have a lot of questions about one of the key elements of the european regulatory process the ce technical file. Preparing your ce technical file takes precision and care, as the requirements for data, documentation, and formatting of this document are very specific. Wha o a practical guide iso. Iso 134852016 medical devices a practical guide 7. You are implementing a new qms or updating your existing qms, the advice given in this handbook is relevant. Electronic health records centers for medicare & medicaid. Find health record. Get high level results! Medical record wikipedia. Internetcorkboard has been visited by 1m+ users in the past month.
Kadlec Patient Records
My Medical Record Opt Out
Medical devices unfpa. Medical device with the intention of making the medical device available for use, under his name; whether or not such a medical device is designed and/or manufactured by that person himself or on his behalf by another person(s) ghtfsg1n055definition of terms. Your medical records hhs.Gov. Find fast answers for your question with govtsearches today! Log in myhealthrecord. Govtsearches has been visited by 100k+ users in the past month. White paper device master records and medical device files. Device master records and medical device files how do they compare? Fda requires the use of a device master record (dmr) for medical devices. The iso 134852016 standard; however, now includes a medical device file (mdf) requirement. What are dmrs? What are mdfs? How do they compare? Technical documentation tecnologias sanitarias. Technical documentation page 2/21 vdtuevdocument dn \hoeppner\mp\nb\rec_vdt2\r2_5_15_rev4.Doc the purpose of this recommendation is to provide guidance to notified bodies, competent authorities and manufacturers on the technical documentation needed to meet the requirements of the medical devices directives.
Electronic Media Job In Delhi
New chapter 4.2.3 medical device file the quality forum online. Hi there, in the 2016 revision, there is a new chapter 4.2.3 medical device file. My company is not producing any medical device, but only components that may be used on a medical device (that's why we are iso 13485 certified, our customers are medical device manufacturer). Health records online now directhit. Also try. Healthcare records. Healthcare records govtsearches. Health record as used in the uk, a health record is a collection of clinical information pertaining to a patient's physical and mental health, compiled from different sources. Australian regulatory guidelines for medical devices. • The therapeutic goods administration (tga) is part of the australian government department of health and ageing, and is responsible for regulating medicines and medical devices. Iso 134852016 how to comply with medical device files. The requirements for medical device files in iso 134852016 are an endeavor by the iso technical committee (tc 210) to create consistent operations for medical device manufacturers, and also to make their quality management systems compliant with the rules of various regulatory bodies. An electronic health record (ehr) is an electronic version of a patients medical history, that is maintained by the provider over time, and may include all of the key administrative clinical data relevant to that persons care under a particular provider, including demographics, progress notes, problems, medications, vital signs, past medical history. Medical devices unfpa. Medical device with the intention of making the medical device available for use, under his name; whether or not such a medical device is designed and/or manufactured by that person himself or on his behalf by another person(s) ghtfsg1n055definition of terms. Health records online now directhit. The service is an online service designed to allow you to communicate with your medical care providers. You can send secure messages to your provider, request an appointment, check on your lab results, view your health record, request a prescription refill, complete registration and health information forms, and read patient education.
Medical Records Technician Function
Health record video results. Find health record if you are looking now.